Ammonia (NH3)-Calibration Gas
Author: ChéAna Morgan
Download the article here: https://egasdepot.com/content/Ammonia (NH3) - Calibration Gas.pdf
Ammonia (NH3)
Ammonia is a colorless gas that has an overwhelming, and immediately suffocating odor. When alone it does not have corrosive properties and it is also not considered to be flammable, but has the potential to cause an explosion when coming into contact with gases such as chlorine, where it then forms NCl3, which is a highly explosive chemical compound. NH3 is most commonly known for its use in household cleaning products as well as its role in “smelling salts” which are used in the medical field and even sports to trigger consciousness in an unconscious person. Ammonia is produced naturally in nature through the decomposition of organic materials such as plants and animals and also exists naturally in humans. Ammonia is used in many different industries to include the pharmaceutical, agriculture, plastics, explosives and pesticides in both its liquid and gas (most dangerous) form. When exposure to high concentrations of Ammonia (NH3) occurs there can be serious if not fatal consequences, most often including respiratory complications caused from inhalation of the gas or vapors. Ensuring you have the proper gas detection systems and calibration gas to ensure it is working properly could mean the difference of life or death.
Also Known As:
- Hydrogen nitride
- Trihydrogen Nitride
- Nitrogen Trihydride
Health Effects of Exposure to Ammonia (NH3) Gas
Ammonia is considered to an extremely hazardous substance and is governed by firm regulations regarding reporting of its use by facilities that produce and/ or store it in abundant quantities. There are three different ways that a person may be affected by the exposure to Ammonia (NH3) gas. The most common way includes inhalation followed by skin/eye contact followed by ingestion. At levels above 50ppm, Ammonia becomes very easy to detect by smell which should give you plenty of time to remove yourself before levels become too dangerous. Once inhalation occurs at high concentrations or Ammonia is inhaled on a consistent basis over time, the gas can cause olfactory paralysis (the loss of smell). This will result in burning of mucous membranes in the nose, throat, and respiratory tract which could lead to respiratory failure or. Severe injury such as burns can occur if you were to come into contact with liquid ammonia, and blindness could occur if it comes into contact with your eyes. Contact with liquefied Ammonia (NH3) has the potential to cause frostbite. Corrosive damage to the mouth, throat and digestive tract could occur if Ammonia was to be ingested.
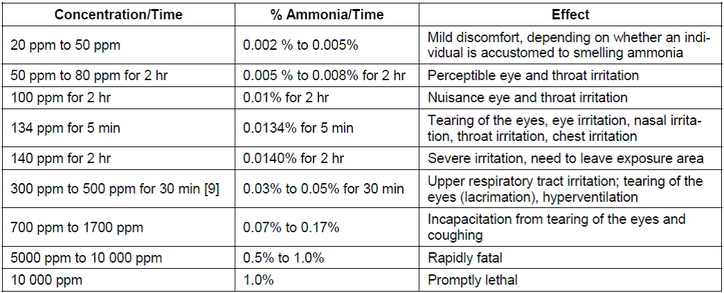

*There is an 8-hour exposure limit of 25 ppm and a short-term (15-minute) exposure limit of 35 ppm for ammonia in the workplace set by OSHA. NIOSH highly recommends that the level in work space air be limited to 50 ppm for 5 minutes of exposure.
Industrial Applications for Ammonia (NH3)
The major players for the use of Ammonia include the agricultural industry where nearly 80% of the total of ammonia (NH3) produced in the United States is accounted for. Ammonia is an essential part in the growth of plants and is often used in the production of fertilizer. You can also find Ammonia in the water and wastewater treatment industry as well as the food and beverage industry where it is used as a source of nitrogen to fuel yeast and microorganisms. Very faint solutions of ammonia are used in the household and commercial cleaning products. Other uses include
Handling & Storage of Ammonia (NH3) Gas
Ammonia is a compressed gas with corrosive properties when in the presence of moisture and the potential to become and explosion hazard when it forms compounds with elements such as chlorine and iodine. Handling Ammonia(NH3) gas cylinders should be done with the utmost care ensuring to never handle it while alone. Never lift a cylinder by the neck ring and avoid contact with other cylinders. Compressed gas cylinders should always be secured appropriately with some type of restraint. Always store cylinders in a cool dry storage area and keep out of direct sunlight. As always, you should read and understand the MSDS associated with the specific gas you are working with to help prevent emergencies from occurring.
Ammonia (NH3) Calibration Gas Cylinders
Egas Depot Offers a wide range of Ammonia (NH3) Calibration Gas Cylinders. With 9 different liter sizes and 7 different options for your ppm requirements available to you, we are a one-stop shop for all of your field calibration needs.

